Which Statement Accurately Describes Isomers
eight.half-dozen: Resonance Structures
- Page ID
- 21750
- To sympathize the concept of resonance.
Resonance structures are a fix of two or more Lewis Structures that collectively draw the electronic bonding of a single polyatomic species including fractional bonds and partial charges. Resonance structures are capable of describing delocalized electrons that cannot exist expressed past a single Lewis formula with an integral number of covalent bonds.
Sometimes i Lewis Structure is not Plenty
Sometimes, fifty-fifty when formal charges are considered, the bonding in some molecules or ions cannot exist described past a single Lewis structure. Resonance is a mode of describing delocalized electrons within sure molecules or polyatomic ions where the bonding cannot be expressed by a single Lewis formula. A molecule or ion with such delocalized electrons is represented by several contributing structures (also called resonance structures or canonical forms). Such is the case for ozone (\(\ce{O3}\)), an allotrope of oxygen with a V-shaped construction and an O–O–O angle of 117.5°.
Ozone (\(O_3\))
1. We know that ozone has a V-shaped structure, then one O atom is fundamental:

two. Each O atom has 6 valence electrons, for a total of 18 valence electrons.
three. Assigning i bonding pair of electrons to each oxygen–oxygen bond gives
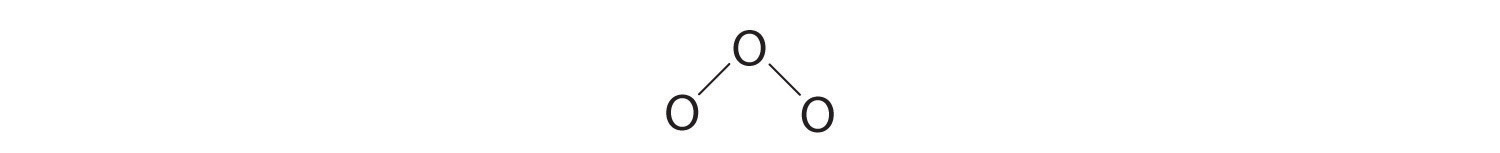
with fourteen electrons left over.
iv. If we identify three lonely pairs of electrons on each final oxygen, we obtain

and have ii electrons left over.
five. At this point, both terminal oxygen atoms have octets of electrons. Nosotros therefore identify the last ii electrons on the primal atom:
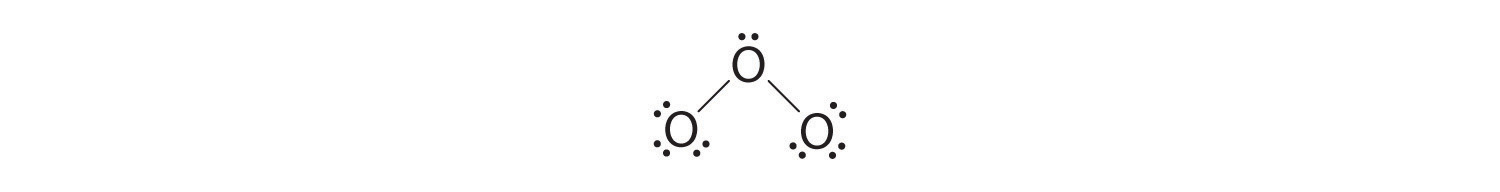
vi. The cardinal oxygen has but 6 electrons. We must convert one lone pair on a terminal oxygen atom to a bonding pair of electrons—merely which one? Depending on which one nosotros choose, we obtain either
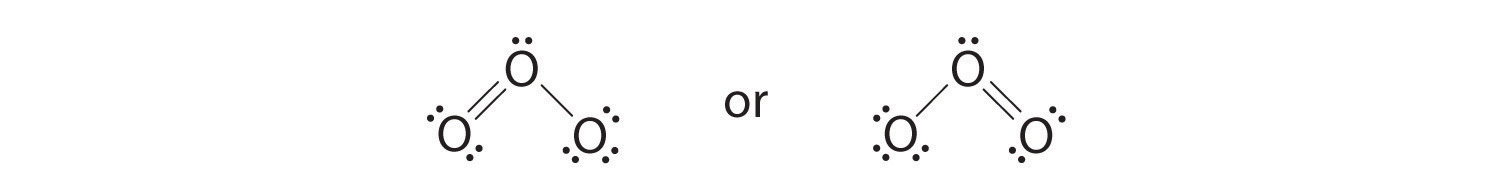
Which is right? In fact, neither is right. Both predict one O–O unmarried bond and 1 O=O double bond. Every bit you will acquire, if the bonds were of unlike types (1 single and one double, for example), they would have different lengths. It turns out, still, that both O–O bail distances are identical, 127.2 pm, which is shorter than a typical O–O single bail (148 pm) and longer than the O=O double bond in O2 (120.seven pm).
Equivalent Lewis dot structures, such equally those of ozone, are called resonance structures. The position of the atoms is the same in the various resonance structures of a compound, but the position of the electrons is different. Double-headed arrows link the different resonance structures of a compound:
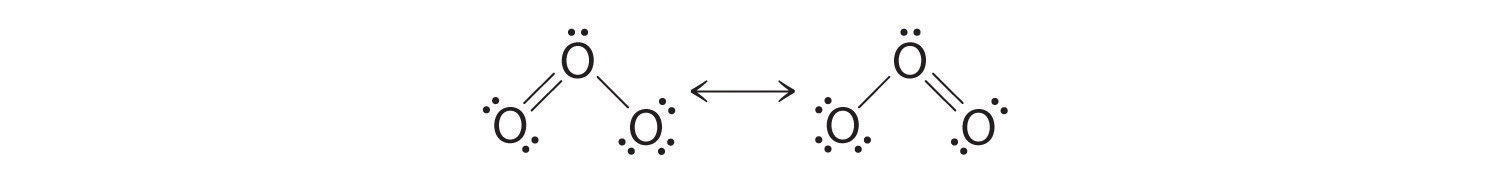
The double-headed arrow indicates that the bodily electronic construction is an average of those shown, not that the molecule oscillates betwixt the two structures.
When it is possible to write more than ane equivalent resonance structure for a molecule or ion, the actual structure is the average of the resonance structures.
The Carbonate (\(CO_3^{two−} \)) Ion
Similar ozone, the electronic construction of the carbonate ion cannot be described by a single Lewis electron construction. Different O3, though, the actual construction of CO3 two − is an average of three resonance structures.
1. Considering carbon is the least electronegative element, we place it in the fundamental position:
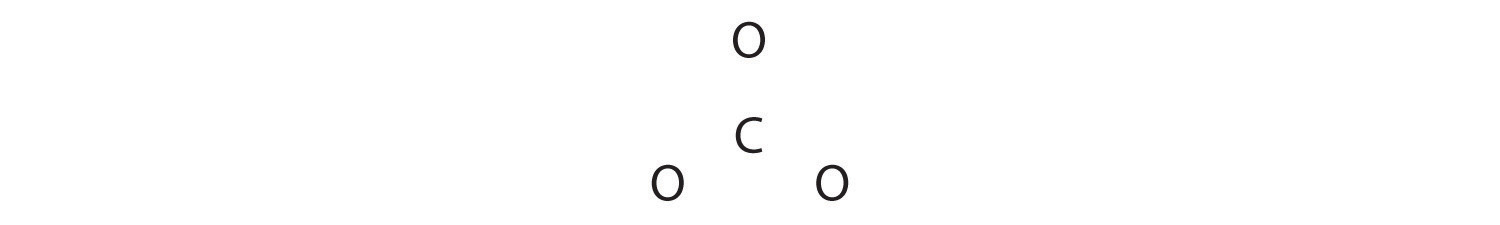
2. Carbon has 4 valence electrons, each oxygen has 6 valence electrons, and there are two more than for the −2 charge. This gives 4 + (3 × vi) + 2 = 24 valence electrons.
three. Six electrons are used to form three bonding pairs between the oxygen atoms and the carbon:
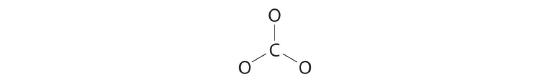
4. We split the remaining eighteen electrons as amid the iii oxygen atoms past placing three lone pairs on each and indicating the −ii accuse:
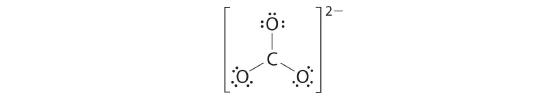
5. No electrons are left for the central cantlet.
vi. At this signal, the carbon atom has merely half dozen valence electrons, then we must take one lone pair from an oxygen and use it to form a carbon–oxygen double bond. In this case, however, at that place are three possible choices:
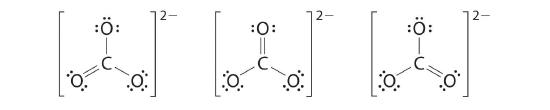
Equally with ozone, none of these structures describes the bonding exactly. Each predicts 1 carbon–oxygen double bond and two carbon–oxygen single bonds, but experimentally all C–O bond lengths are identical. We can write resonance structures (in this case, 3 of them) for the carbonate ion:
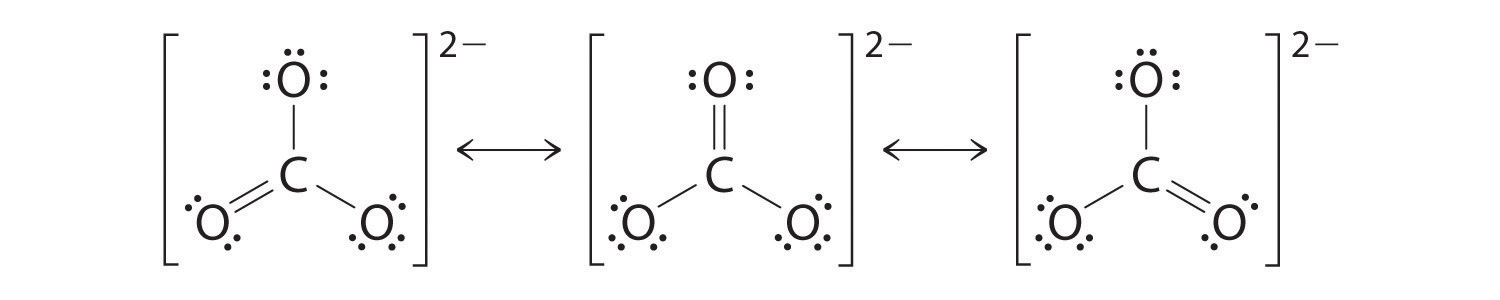
The actual construction is an average of these three resonance structures.
The Nitrate (\(NO_3^-\)) ion
i. Count up the valence electrons: (1*5) + (3*6) + 1(ion) = 24 electrons
2. Depict the bond connectivities:
3. Add octet electrons to the atoms bonded to the centre atom:
4. Place whatsoever leftover electrons (24-24 = 0) on the center atom:
five. Does the central atom have an octet?
- NO, information technology has 6 electrons
- Add together a multiple bond (start try a double bond) to see if the fundamental atom tin can reach an octet:
6. Does the central cantlet accept an octet?
- YES
- Are at that place possible resonance structures? YES
Note: We would expect that the bail lengths in the \(\ce{NO_3^{-}}\) ion to be somewhat shorter than a single bond.
Benzene is a common organic solvent that was previously used in gasoline; it is no longer used for this purpose, notwithstanding, because it is now known to exist a carcinogen. The benzene molecule (\(\ce{C6H6}\)) consists of a regular hexagon of carbon atoms, each of which is also bonded to a hydrogen atom. Use resonance structures to describe the bonding in benzene.
Given: molecular formula and molecular geometry
Asked for: resonance structures
Strategy:
- Describe a construction for benzene illustrating the bonded atoms. And so calculate the number of valence electrons used in this drawing.
- Subtract this number from the full number of valence electrons in benzene and and so locate the remaining electrons such that each atom in the structure reaches an octet.
- Depict the resonance structures for benzene.
Solution:
A Each hydrogen atom contributes 1 valence electron, and each carbon atom contributes 4 valence electrons, for a total of (6 × 1) + (six × 4) = xxx valence electrons. If we identify a single bonding electron pair betwixt each pair of carbon atoms and between each carbon and a hydrogen atom, we obtain the following:
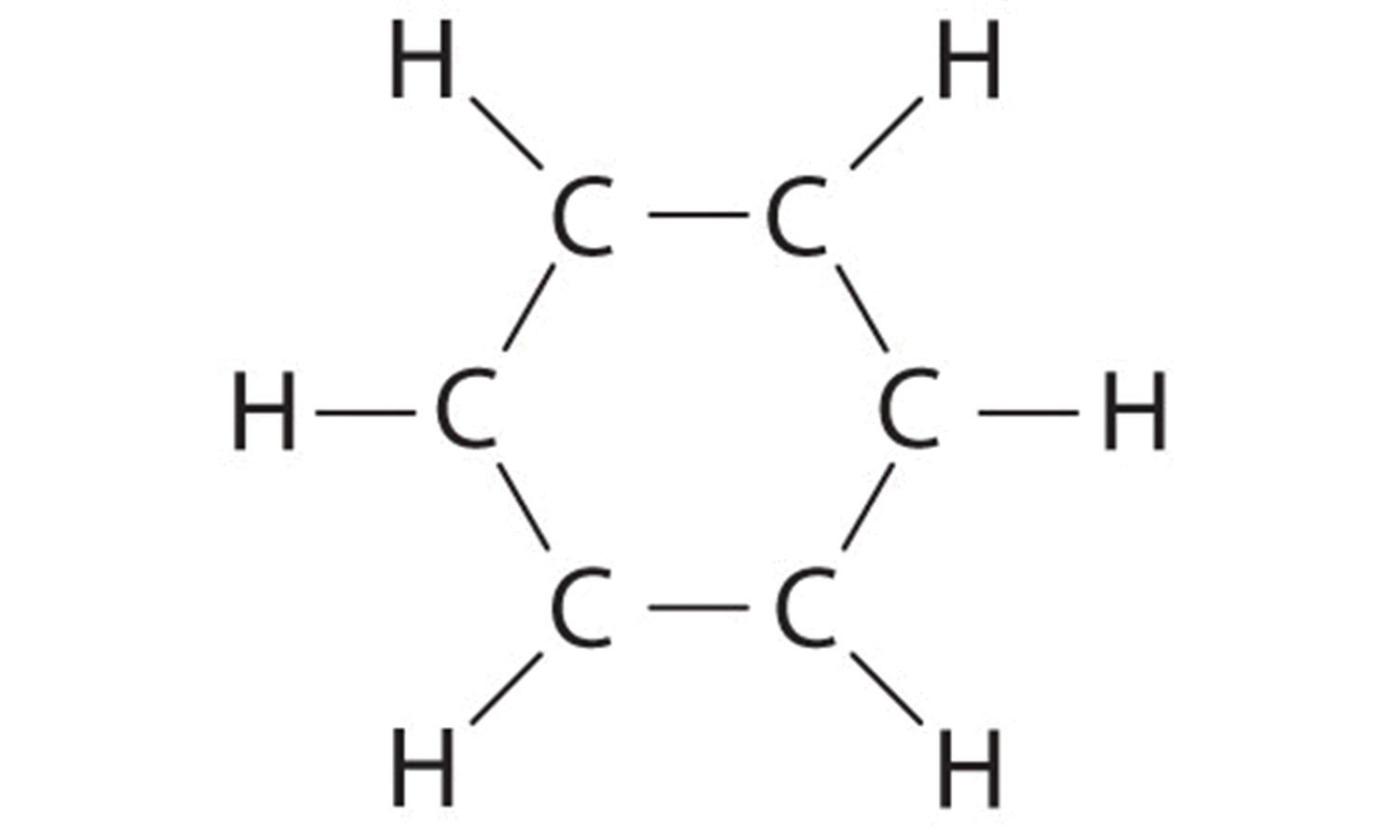
Each carbon atom in this structure has only six electrons and has a formal charge of +1, only we accept used simply 24 of the 30 valence electrons.
B If the half dozen remaining electrons are uniformly distributed pairwise on alternate carbon atoms, nosotros obtain the following:
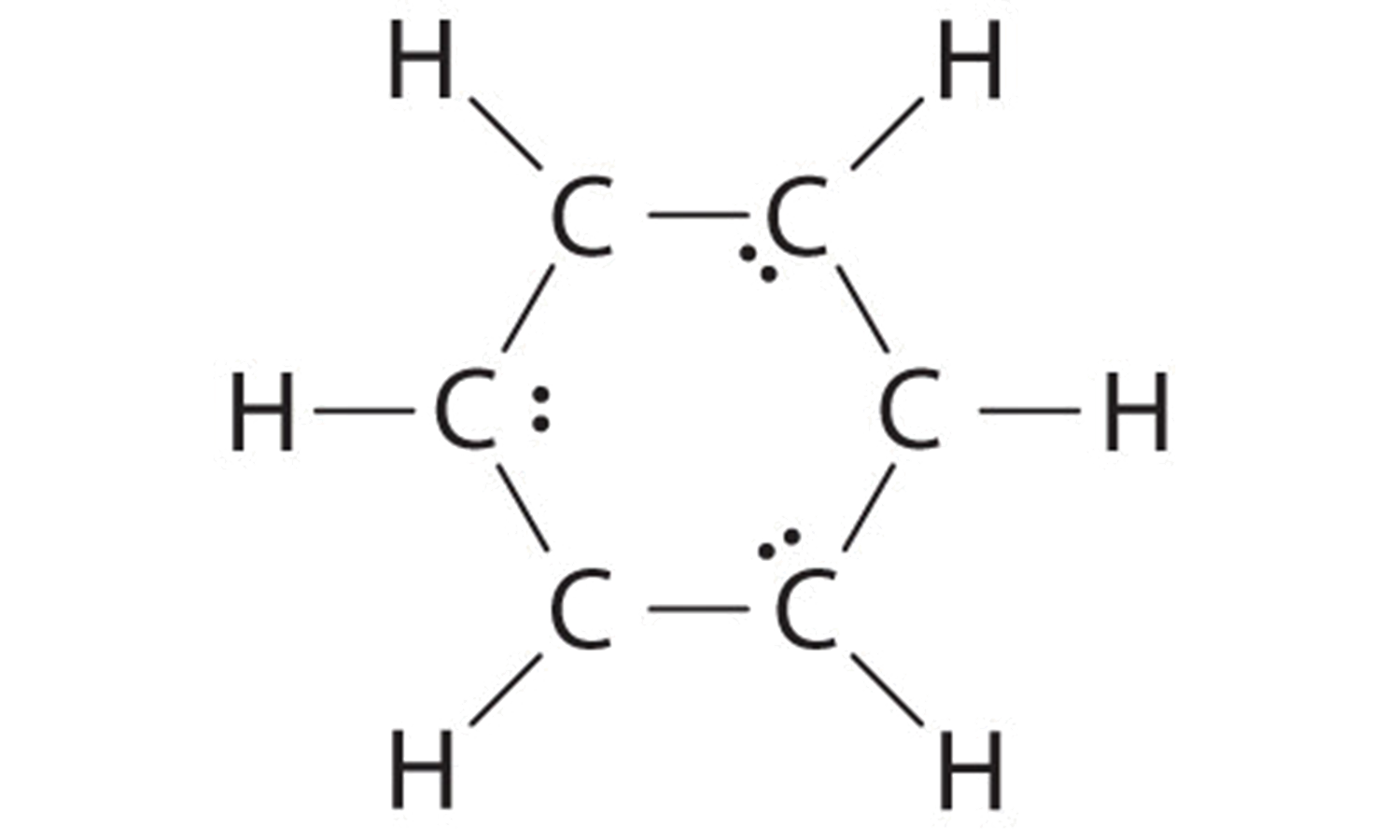
Three carbon atoms now have an octet configuration and a formal charge of −1, while three carbon atoms have only 6 electrons and a formal accuse of +1. We tin can convert each lonely pair to a bonding electron pair, which gives each atom an octet of electrons and a formal charge of 0, by making three C=C double bonds.
C In that location are, however, two ways to practise this:
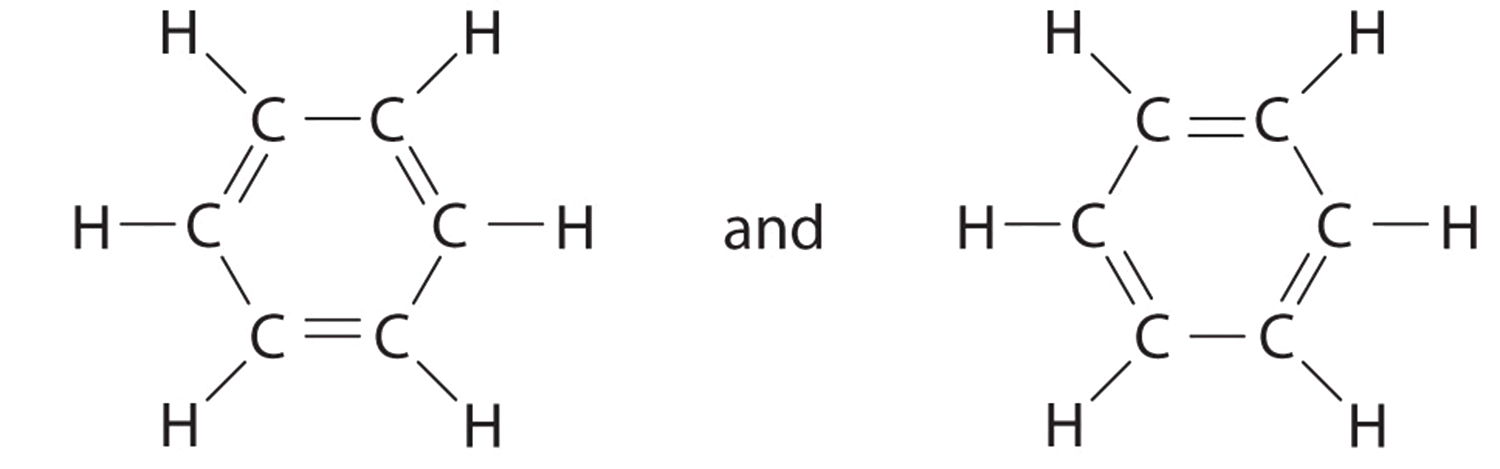
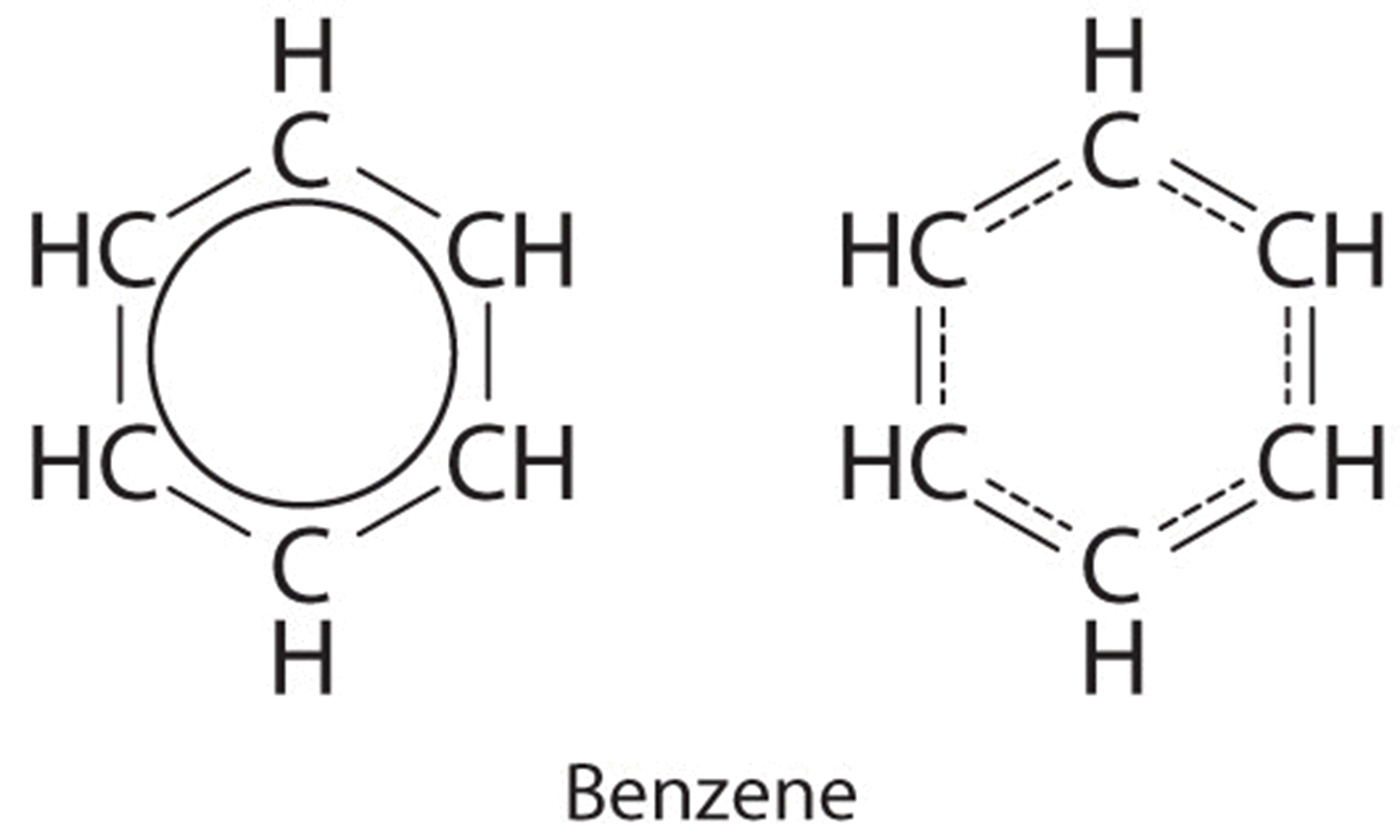
Each construction has alternating double and single bonds, but experimentation shows that each carbon–carbon bail in benzene is identical, with bond lengths (139.9 pm) intermediate betwixt those typically establish for a C–C single bail (154 pm) and a C=C double bond (134 pm). We can describe the bonding in benzene using the two resonance structures, just the actual electronic structure is an boilerplate of the two. The beingness of multiple resonance structures for aromatic hydrocarbons like benzene is ofttimes indicated by drawing either a circumvolve or dashed lines inside the hexagon:
The sodium salt of nitrite is used to save muscle spasms. Draw two resonance structures for the nitrite ion (NO2 −).
- Answer
-
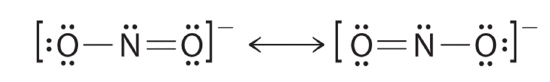
There is a double bond between one oxygen and the nitrogen, It can exist found on the left oxygen or the correct oxygen. Resonance structures are specially common in oxoanions of the p-block elements, such every bit sulfate and phosphate, and in aromatic hydrocarbons, such as benzene and naphthalene.
If several reasonable resonance forms for a molecule exists, the "actual electronic structure" of the molecule will probably be intermediate between all the forms that yous can depict. The archetype example is benzene in Example \(\PageIndex{1}\). One would look the double bonds to be shorter than the single bonds, only if one overlays the two structures, y'all see that one structure has a single bond where the other structure has a double bond. The best measurements that nosotros tin brand of benzene do not show two bond lengths - instead, they show that the bond length is intermediate between the two resonance structures.
Resonance structures is a mechanism that allows us to use all of the possible resonance structures to try to predict what the actual form of the molecule would be. Single bonds, double bonds, triple bonds, +one charges, -1 charges, these are our limitations in explaining the structures, and the true forms tin can be in between - a carbon-carbon bond could be mostly single bond with a little bit of double bond grapheme and a partial negative charge, for instance.
Summary
Some molecules accept two or more chemically equivalent Lewis electron structures, called resonance structures. Resonance is a mental exercise and method within the Valence Bail Theory of bonding that describes the delocalization of electrons inside molecules. These structures are written with a double-headed arrow between them, indicating that none of the Lewis structures accurately describes the bonding just that the actual structure is an average of the private resonance structures. Resonance structures are used when 1 Lewis structure for a unmarried molecule cannot fully describe the bonding that takes place between neighboring atoms relative to the empirical information for the actual bond lengths between those atoms. The cyberspace sum of valid resonance structures is defined as a resonance hybrid, which represents the overall delocalization of electrons within the molecule. A molecule that has several resonance structures is more than stable than one with fewer. Some resonance structures are more favorable than others.
Which Statement Accurately Describes Isomers,
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/08%3A_Basic_Concepts_of_Chemical_Bonding/8.06%3A_Resonance_Structures
Posted by: joneshaters.blogspot.com


0 Response to "Which Statement Accurately Describes Isomers"
Post a Comment