Is Cs2 Soluble In Water
What is Carbon Disulfide?
CS2 is an organosulfur compound and a volatile liquid with the chemic name carbon Disulfide. It is besides called Carbon bisulfide or disulfidocarbon or methanedithione.
Tabular array of Contents
- Carbon Disulfide Structure
- Properties of Carbon Disulfide – CS2
- CS2 Uses
- Carbon Disulfide Reactions
- Health Hazards
- Frequently Asked Questions
Carbon Disulfide is a solvent for sulfur, bromine, fats, rubber, phosphorus, asphalt, selenium, iodine, and resins. Information technology has been widely used to purify single-walled carbon nanotubes and in the manufacturing of flotation agents.
Carbon disulfide is a flammable, colourless to light yellow, poisonous, volatile liquid which has a strong disagreeable smell. It has a flash bespeak value of -22°F and is insoluble in water. It is denser than water, therefore, sinks in it.
Carbon Disulfide Structure – CStwo
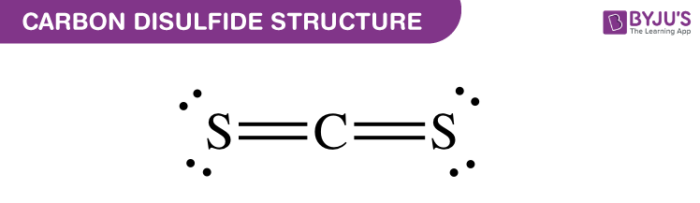
Properties of Carbon Disulfide – CS2
| CStwo | Carbon Disulfide |
| Molecular weight of CS2 | 76.thirteen g/mol |
| Density of Carbon Disulfide | ane.539 chiliad/cm3 |
| Melting point of Carbon Disulfide | −111.61 °C |
| Humid betoken of Carbon Disulfide | 46.24 °C |
CSii Uses (Carbon Disulfide)
- Carbon Disulfide is used in the production of carbon tetrachloride.
- Used as preparing soil disinfectants.
- Used in the manufacturing of rayon.
- Used as a solvent for iodine, phosphorous, etc.
- Used to industry electronic vacuum tubes.
- Used as a solvent in condom-making industries.
- Used in camphor.
- Used in generating petroleum catalysts.
- Used as pesticide intermediate.
Carbon Disulfide Reactions
-
- Reacts with oxygen produce carbon dioxide and sulfur dioxide.
CS2 + iii O2 → CO2 + 2 So2
-
- Sodium sulfide undergoes a reaction to produce trithiocarbonate:
NatwoDue south + CS2 → [Na+]2[CS3 2−]
-
- Carbon disulfide is reduced with sodium to produce sodium 1,three-dithiole-two-thione-four,five-dithiolate and sodium trithiocarbonate.
4 Na + four CSii → NatwoC3Sv + NatwoCS3
-
- Carbon tetrachloride is produced by chlorination of CS2
CS2 + three Cl2 → CCl4 + S2Cl2
Health Hazards
Information technology affects the CNS, eyes, liver, skin, cardiovascular organisation, and kidneys. Information technology is absorbed through the pare in its liquid or vapour grade, ingested or inhaled. The probable oral lethal dose for humans is betwixt 0.5 to 5 grams kilogram per person. Also, it leads to disturbance of vision and sensory organs. The lowest lethal dose for a human is reported every bit fourteen mg/kg per person.
Oft Asked Questions
Is carbon disulfide an acid?
Carbon disulfide with the formula CS2 is a colourless volatile liquid. The compound is commonly used in organic chemistry equally a edifice cake, too as a non-polar industrial and chemical solvent. This has an "ether-like" odour but commonly, industrial samples are polluted with impurities that smell foul.
Where does carbon disulfide come up from?
Express quantities of carbon disulfide are present in nature in gasses emitted to the world's surface, e.yard. in volcanic eruptions or over marshes. Microorganisms may as well create carbon disulfide-containing gas in soil. Commercial carbon disulfide is produced at very high temperatures by adding carbon and sulphur.
Is carbon disulfide made of atoms or molecules?
Carbon disulphide is an organic chemical compound or molecule made up of ane carbon and two sulphur atoms. The smallest particles which accept complete backdrop of carbon disulphide are molecules, not atoms.
What intermolecular forces does carbon disulfide have?
Due to the departure in electronegativities between C and S, the C-Southward bond is nonpolar. Carbon disulphide has a linear construction in which ii bond dipoles are in opposite directions resulting in cancelling out the dipole moments. So carbon disulphide is non-polar.
Learn more well-nigh the Construction, physical and chemical properties of CS2 from the experts at BYJU'Due south.
Is Cs2 Soluble In Water,
Source: https://byjus.com/chemistry/carbon-disulfide/
Posted by: joneshaters.blogspot.com


0 Response to "Is Cs2 Soluble In Water"
Post a Comment